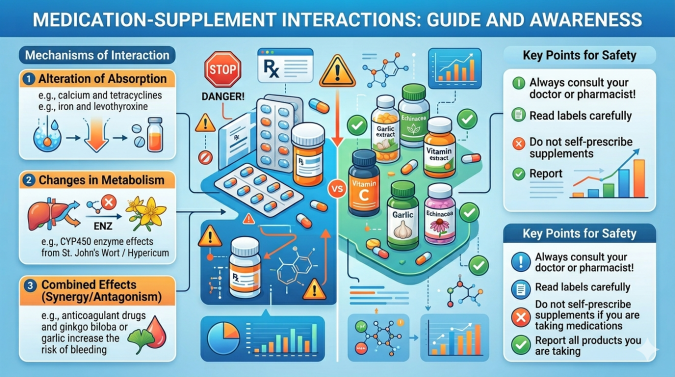
Many patients rely on supplements to cure common disorders, like headaches, joint pain, and fatigue. But are these remedies really beneficial? Supplements may be helpful when taken alone, but the concurrent use of drugs or prescribed medications could entail some life-threatening risks. This article explores the hot topic of interactions between popular supplements and medications. Let’s start with understanding what supplements are.
Supplements Overview
Dietary or nutritional supplements are a category of complementary products intended to supplement the diet. They include vitamins, amino acids, enzymes, and herbal extracts. Originally used to address nutritional deficiencies, these products are now largely used as additional medications to treat several clinical conditions, such as diabetes or high blood pressure. Sometimes they are prescribed by doctors, but due to advertising and consumer perception, they are usually bought without a medical prescription.
Most people, in fact, perceive supplements as harmless or, rather, extremely beneficial, nearly as a miraculous medicine. But reality is, unfortunately, fairly different.
Supplements, indeed, can interact with several kinds of medications, like antidepressants and anticoagulants. Moreover, these interactions may cause organ damage.
Why Do Supplements and Drugs Interact?
Often, supplements have the same effects as the drugs one is taking. These interactions overlap and are not clearly communicated to patients.
The main interactions occur because supplements can inhibit the enzymes that metabolize a wide range of prescription drugs. One of the most clamorous cases is with berberine.
The Berberine Case
Berberine is a popular organic supplement also known as “nature Ozempic.” Derived from plants, it is an alkaloid with digestive and antimicrobial properties. Studies also found that berberine supports metabolic health and lowers blood sugar. For this reason, many patients use it as a supplement to lose weight. But the studies showing whether berberine could replace GLP-1 medication (Ozempic) to lose weight are still inconclusive.
Moreover, berberine and GLP-1 don’t act on the same receptors, and this detail could make the concurrent usage of the supplement and the medication highly risky.
The most dangerous scenario happened to a transplanted 16-year-old girl, where berberine interacted with tacrolimus, the immunosuppressant used in organ transplants. The supplement increased the immunosuppressant to a toxic level, causing the girl to develop kidney failure.
The interaction occurred because berberine is a strong inhibitor of the CYP3A4 and CYP3A5 enzymes in the liver and small intestine, as well as the drug transporter P-glycoprotein (P-gp). Since tacrolimus is metabolized by CYP3A4/5, its inhibition by berberine leads to reduced clearance of tacrolimus and significantly increased blood concentration (toxicity).
Other case reports relate to old transplanted patients. The interaction between berberine and tacrolimus is always the same: renal toxicity.
The Omega-3 Supplements Case
Another case involves omega-3 supplements. A post published via LinkedIn revealed that oil fish supplements may damage the brain when taken at high doses. Experts emphasize that current evidence does not confirm direct harm to the brain from fish oil supplements, but highlights the importance of balanced dosing and medical guidance. Overconsumption of any supplement may disrupt natural physiological processes, which is why personalized nutrition is increasingly recommended in modern healthcare practices today globally.
Supplements and Chronic Diseases
The risk of interaction between supplements and drugs is higher in the case of chronic diseases. However, according to recent estimations, between 40% and 60% of U.S. adults with chronic disease take dietary supplements, and an estimated 25% takes supplements along with prescription drugs.
The greatest concerns about harmful interactions involve herbal supplements. Herbal extracts, indeed, may interact with drugs at the pharmacokinetic and pharmacodynamic levels. A pharmacokinetic interaction occurs when supplements change the blood concentration of drugs, while a pharmacodynamic interaction occurs when supplements have a direct pharmacological action unrelated to drug intake.
The popular curcumin, for instance, an active compound extracted from turmeric, is appreciated for its anti-inflammatory properties. Still, a study showed that it induces CYP1A2, which could lead to decreased levels of many antidepressant and antipsychotic medications.
Sometimes, the pharmacodynamic effects of herbal supplements may be more detrimental than the pharmacokinetic ones.
It happened to me when I decided to take ashwagandha powder to mitigate the symptoms of my autoimmune thyroiditis and the early symptoms of my Parkinson’s. At the time, I hadn’t taken Levodopa yet.
I added a tablespoon of the powder to a cup of fruit juice in the morning, but after three weeks of the intake, I experienced an awful sense of fatigue and a worsening of my motor symptoms.
In the following days, a careful medical check with an endocrinologist and a neurologist reported that ashwagandha powder suppressed the adrenal glands, causing extreme fatigue and exacerbating the progression of my neurological disease.
Today, I still take nutritional supplements, but they are prescribed by my doctors with balanced doses and at a far distance from the medications I must take for my chronic diseases.
Currently, my array of prescribed supplements includes vitamin D pills for Hashimoto’s thyroiditis, vitamin B effervescent tablets for the mental and physical fatigue induced by Parkinson’s, and Ginkgo biloba as a natural anticoagulant. The only prescribed drugs to treat my chronic diseases are Sinemet and Vortioxetine.
As regards Sinemet, aka levodopa, I have another firsthand experience with supplement interactions. Usually, I tend to use propolis extract to cure a sore throat. But, a few months ago, I noticed that the intake of Sinemet and propolis caused me motor symptoms, because the herbal extract impeded the absorption of levodopa.
Today, when I have other concurrent diseases, such as fever, tooth pain, or the flu, I always call my doctor to avoid interactions between the supplements I take and the new prescribed medication to cure seasonal diseases.
How to Verify Supplement and Drug Interactions
So far, it has not been easy for clinicians to verify the side effects of supplements. These products are often sold as consumer goods. In several countries, you can also buy them at the supermarket. This happens because regulatory agencies do not label supplements as official medications, exonerating the producer from including the product information leaflet. Even when the latter is present, it fails to indicate any interaction with drugs.
A practical way to check these interactions and plan individualized prescriptions for patients is through online databases known as safety checkers or safety databases.
Safety checkers contain alphabetical and ordered information on all the types of available supplements, their side effects, and interactions with drugs. Their clinical details are science and research-backed.
The main information of online safety checkers includes:
Interactions: Which supplements, drugs, and drug classes are contraindicated. Each interaction is tagged with severity and strength of evidence for easy risk evaluation.
Side effects: Unintended or undesirable effects of supplement use.
Precautions: Warnings for specific demographics, populations, and conditions.
Pregnancy and lactation: Dedicated guidance for these critical life stages.
Quality concerns: Contamination or adulteration issues.
WADA: Whether the supplement is prohibited by the World Anti-Doping Agency.
Quality Concerns
Getting information about the quality of a supplement is of vital importance to prescribe a safe and effective product and minimize interaction risks. The safety database of Examine, for example, related to my search on Ginkgo biloba reported the following concerns: A study that analyzed Ginkgo biloba supplements from Europe found that around 80% did not contain the active metabolites in the amounts stated on the labels. Another study that analyzed Ginkgo biloba supplements from the United States found that some products contained a dangerous amount (up to 90,000 parts per million, compared to the safe limit of 5 parts per million) of ginkgolic acids (a potentially toxic compound naturally found in Ginkgo biloba). It also showed evidence of adulteration through substitution with other plant materials or the addition of flavonoids from other sources, or the supplement contained doses significantly lower than declared.
Conclusion
Supplements, hence, are not as safe as one could believe. Much of their harmful effects depend on the wrong dosage, but also on the purity of the source from which they are extracted and from production processes, which vary from country to country. A safety check before prescribing them could make a difference between patient safety and clinical risk.